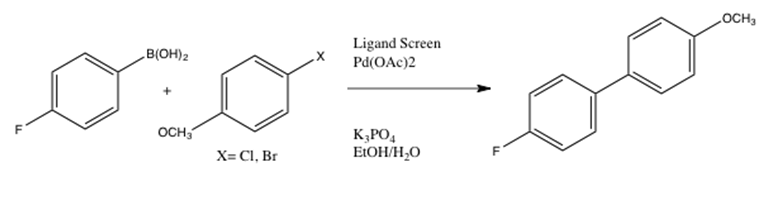
Highly recoverable, reusable, cost-effective, and Schiff base functionalized pectin supported Pd(II) catalyst for microwave-accelerated Suzuki cross-coupling reactions - ScienceDirect

Amazon.com: Suzuki-Miyaura Cross-Coupling Reaction and Potential Applications: 9783038425564: Kostas, Ioannis D: ספרים

A Bulky Chiral N-Heterocyclic Carbene Palladium Catalyst Enables Highly Enantioselective Suzuki–Miyaura Cross-Coupling Reactions for the Synthesis of Biaryl Atropisomers | Journal of the American Chemical Society

Mechanistic Aspects of the Palladium‐Catalyzed Suzuki‐Miyaura Cross‐Coupling Reaction - D'Alterio - 2021 - Chemistry – A European Journal - Wiley Online Library

A Bulky Chiral N-Heterocyclic Carbene Palladium Catalyst Enables Highly Enantioselective Suzuki–Miyaura Cross-Coupling Reactions for the Synthesis of Biaryl Atropisomers | Journal of the American Chemical Society

Palladium catalyzed asymmetric Suzuki–Miyaura coupling reactions to axially chiral biaryl compounds: Chiral ligands and recent advances - ScienceDirect

Mechanistic Aspects of the Palladium‐Catalyzed Suzuki‐Miyaura Cross‐Coupling Reaction - D'Alterio - 2021 - Chemistry – A European Journal - Wiley Online Library

Palladium Nanoparticles on Covalent Organic Framework Supports as Catalysts for Suzuki–Miyaura Cross-Coupling Reactions | ACS Applied Nano Materials

Product and side products of the Suzuki-Miyaurac ross coupling using aG... | Download Scientific Diagram

Recent Advances in the Palladium Catalyzed Suzuki–Miyaura Cross-Coupling Reaction in Water | Catalysis Letters

Mechanistic Aspects of the Palladium‐Catalyzed Suzuki‐Miyaura Cross‐Coupling Reaction - D'Alterio - 2021 - Chemistry – A European Journal - Wiley Online Library
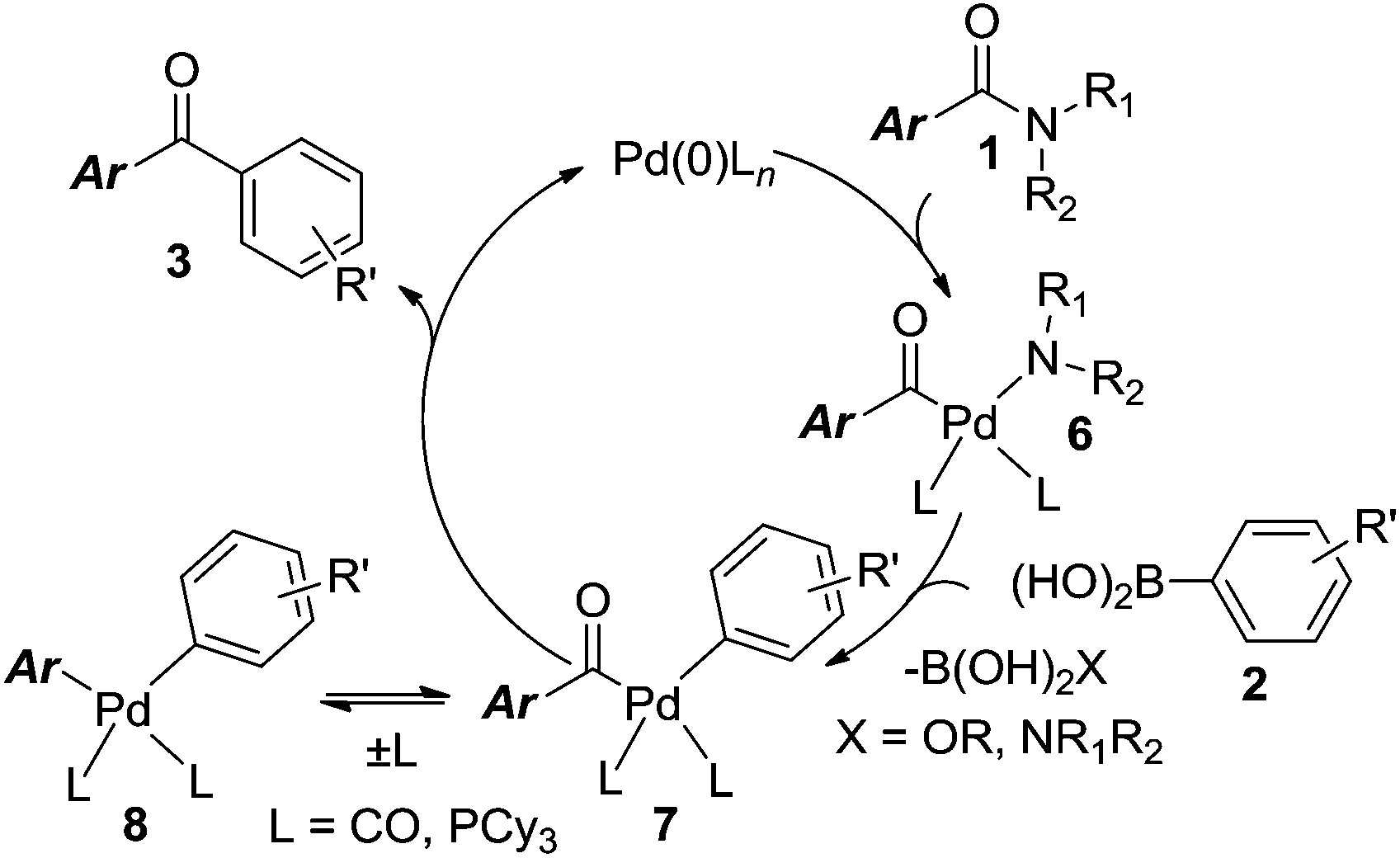
Palladium-catalyzed Suzuki–Miyaura coupling of amides by carbon–nitrogen cleavage: general strategy for amide N–C bond activation - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C6OB00084C

Palladium-Catalyzed Decarbonylative Suzuki–Miyaura Coupling of Amides To Achieve Biaryls via C–N Bond Cleavage | The Journal of Organic Chemistry

Palladium-Catalyzed Suzuki–Miyaura Coupling of Aryl Esters | Journal of the American Chemical Society

Palladium-Catalyzed Suzuki–Miyaura Coupling of Aryl Esters | Journal of the American Chemical Society

Mechanistic Aspects of the Palladium‐Catalyzed Suzuki‐Miyaura Cross‐Coupling Reaction - D'Alterio - 2021 - Chemistry – A European Journal - Wiley Online Library

A Bulky Chiral N-Heterocyclic Carbene Palladium Catalyst Enables Highly Enantioselective Suzuki–Miyaura Cross-Coupling Reactions for the Synthesis of Biaryl Atropisomers | Journal of the American Chemical Society